New Nose Maps
Two new papers on the organization of smell in the brains of mice
A friend sent me a link last week. Two papers, published the same day in Cell, both from Harvard, both presenting a map of smell receptors in the mouse nose that scientists had spent decades assuming didn't exist. Mouse olfactory anatomy sits at some distance from my daily practice, but writing about something forces a depth of understanding that reading alone rarely produces, so here we go again.
When scientists mapped the other senses across the twentieth century, they found spatial order everywhere. The cochlea encodes sound frequency along its length, low tones at one end and high at the other, and that organization carries through into the auditory cortex. The visual system maps the visual field onto the retina and then again onto the brain. Touch follows a similar logic. Olfaction held out as the exception.
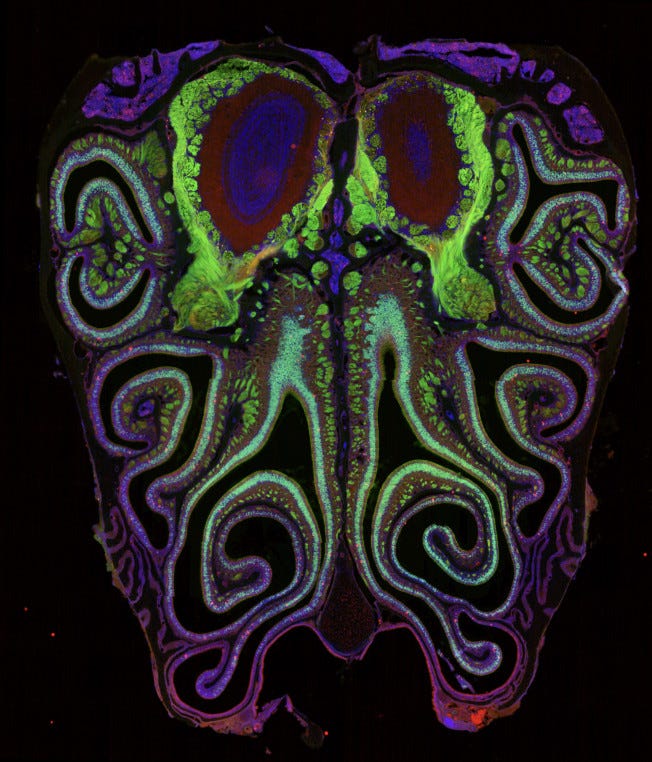
The mouse nose contains around 20 million olfactory sensory neurons, each expressing exactly one receptor type out of roughly 1,100 total. Scientists first identified smell receptor genes in 1991 and spent the following three decades trying to determine whether those 1,100 types had any spatial order within the olfactory epithelium, the sheet of tissue lining the nasal cavity where detection happens. They concluded: not really. A handful of broad anatomical zones provided loose constraints, but within each zone, which receptor a neuron expressed and where it sat seemed essentially random. The accepted model held that smell operated through a fundamentally different logic than the other senses, distributed and unordered at the fine scale. Both new papers challenge that directly.
The Datta lab at Harvard Medical School examined approximately 5.5 million cells from more than 300 mice, identifying which receptor each neuron expressed and precisely where in the tissue it sat. The Dulac and Zhuang labs at Harvard used a spatial imaging technology called MERFISH to visualize nearly the entire receptor gene family within intact tissue. Both groups, working independently with different methods, arrived at the same finding.
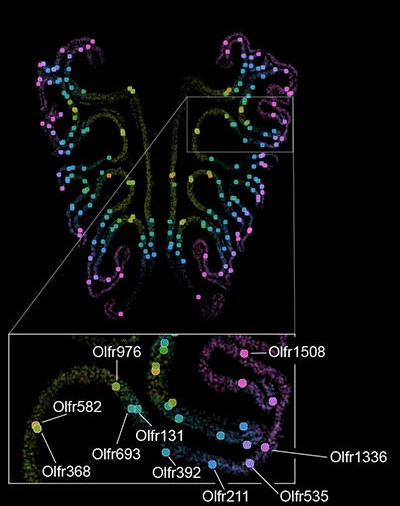
The 1,100 receptor types arrange themselves according to a precise, reproducible spatial logic. Each receptor type occupies a characteristic position along the axis running from the top of the nose toward the bottom, with neurons of the same type clustering at their own mean position and forming overlapping bands. The Dulac/Zhuang paper added that the organization also runs in ring-like gradients expanding from the center of the epithelium outward. The overall picture resembles a coordinate system. Each of the 1,100 receptor types holds a specific address, consistent from animal to animal.
Nose and Brain
The spatial organization extends beyond the nose. Neurons expressing the same receptor type send their axons to the same location in the olfactory bulb, the brain’s first processing station for smell, converging on discrete clusters called glomeruli. The Datta lab found that the map in the nose aligns precisely with this map in the brain. The Dulac/Zhuang paper traced this in three dimensions, showing that the nose’s spatial gradients transform systematically onto the olfactory bulb’s axes.
The Datta lab also identified a partial molecular mechanism, a signaling molecule called retinoic acid exists in a gradient across the developing nose and appears to guide which receptor type a neuron expresses based on its position. This addresses some of how the map forms. It doesn’t yet address why the ordering takes the specific shape it does, or what logic, if any, governs which receptor ends up where.
What This Means for Human Olfaction
Mice serve as a common model for studying olfaction because the basic architecture resembles ours. Both species rely on receptor neurons in a nasal epithelium projecting to an olfactory bulb, but this doesn’t guarantee that every principle discovered in mice transfers to humans. The researchers acknowledged this directly. Human receptor maps don’t exist yet. Whether our epithelium follows the same spatial logic remains open.
The MERFISH technique doesn’t require genetically engineered animals and can work on postmortem human tissue, so further exploration seems likely. Looking at these maps, I find myself curious whether neighboring receptor stripes detect chemically related molecules, and whether proximity in the nose carries any perceptual meaning at all. Those questions sat just out of reach when the system appeared random. Now they’re at least askable, if not directly researchable.
References
Brann, D.H., Tsukahara, T., Tau, C., Kalloor, D., Lubash, R., Thamarai Kannan, L., Klimpert, N., Kollo, M., Escamilla-Del-Arenal, M., Bintu, B., Schaefer, A., Fleischmann, A., Bozza, T., & Datta, S.R. (2026). A spatial code governs olfactory receptor choice and aligns sensory maps in the nose and brain. Cell. https://doi.org/10.1016/j.cell.2026.03.051
Bintu, B., Isogai, Y., Jenie, I., Zhuang, X., & Dulac, C. (2026). Spatial organization and detection of social odors in mouse primary olfactory system. Cell. https://www.cell.com/cell/fulltext/S0092-8674(26)00389-2